Translate this page into:
Wake-Up Sleepyhead: Unilateral Diencephalic Stroke Presenting with Excessive Sleepiness
This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Altered sleep architecture and stroke share a reciprocal relationship. More than half of the stroke patients display sleep abnormalities including hypersomnia, insomnia, parasomnia, periodic limb movements, or sleep-disordered breathing. Conversely, one of the major causes of severe organic hypersomnia is acute brainstem strokes, involving thalamic infarctions, which may be reversible over 6–12 months. Here, we report a patient with increased lethargy and drowsiness who was diagnosed to have a right thalamic and hypothalamic ischemic stroke.
Keywords
Drowsiness with stroke
paramedian thalamic stroke
thalamic infarction
thalamic stroke
INTRODUCTION
Sleep architecture consists of nonrapid eye movement (NREM) and REM components which alternate in 90-min cycles. Sleep disorders, including obstructive sleep apnea, daytime hypersomnolence, and sleep-disordered breathing, are being increasingly identified as causative factors of both cardiovascular and cerebrovascular diseases. Conversely, central nervous system lesions, including strokes, have been reported to disturb sleep cycle, structure, and pattern leading to changes in the electroencephalography (EEG) and polysomnography recordings. The right hemispheric strokes have been shown to depress REM sleep density with an increase in NREM (particularly NREM 1 and 2) as compared to left hemispheric strokes which reduce the slow-wave sleep (SWS) (NREM 4).[1] Diencephalic lesions, including paramedian thalamic infarctions, cause severe hypersomnia with memory and attention deficits, oculomotor palsies, and mild gait ataxia.[2] Although commonly associated with bilateral lesions, unilateral thalamic strokes are being now reported as a frequent cause of hypersomnolence.
CASE REPORT
A 57-year-old right-handed male, previously diagnosed hypertensive, was brought by family members with complaints of being apathetic and having excessive sleepiness for the last 2 days as if he had consumed a bottle full of sleeping pills. Although he could be aroused by minimal stimulus, he preferred to go back to sleep soon. The patient reported that he was not happy with his sleep quality and felt an urge to sleep all the time. There were no other complaints of weakness, sensory symptoms, or memory disturbances. The patient was afebrile, arousable to commands, oriented and was of average built with a body mass index of 25.9 kg/m2. Neurological examination was nonsignificant for any lateralizing deficits. Mini-Mental State Examination score was 20/30 with defects in attention and recall. He was evaluated for causes related to metabolic or septic encephalopathy; however, blood parameters were normal. Magnetic resonance (MR) imaging brain showed an acute diencephalic infarct involving the right anterior and paramedian thalamus and the hypothalamus [Figure 1]. MR angiogram of cerebral vessels was normal. EEG done did not show any epileptiform discharges. Evaluation of sleep revealed an Epworth Sleepiness Score of 18. He was managed conservatively and showed symptomatic improvement with improved arousal during follow-up visit after 3 months.
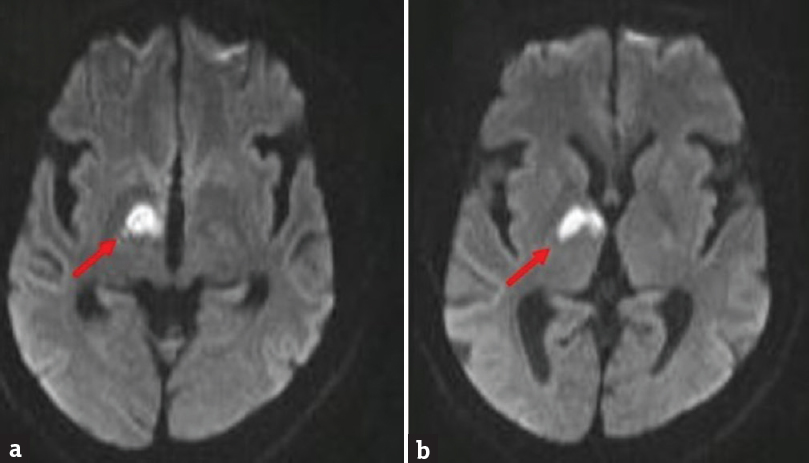
- Magnetic resonance imaging brain diffusion-weighted imaging images (axial cuts) showing acute infarct in the right thalamus involving the anterior and paramedian regions (a) and the hypothalamus (b)
DISCUSSION
Sleep architecture, pattern, and pathology have always been an enigma for physiologists and neurophysicians. Normal sleep cycle consists of alternating NREM and REM phases, which are carefully coordinated by the central nervous system. Normal sleep is essential for homeostatic regulation and stabilization of cardiovascular system,[3] blood pressure,[4] autonomic nervous system,[5] and the central nervous system. Decreased ascending reticular activating system (ARAS) activation, subsequent to brainstem stroke syndromes with the involvement of bilateral paramedian thalamus, thalamomesencephalic region, or medial pontomedullary region produce uncontrolled sleep with diminished arousal.[6]
Thalamus (greek = inner chamber) is a key relay station in the brain with the majority of its nuclei projecting to the cerebral cortex. The dorsal median (DM) nucleus of the thalamus plays an important role in sleep regulation and switching between sleeping and wakeful states. Disruption of the ARAS due to thalamic lesions with involvement of the DM nucleus has been shown to cause dysregulated sleep with a reduction in sleep spindles and SWS. Involvement of the centromedian nucleus along with subthalamic nuclei, as seen in a paramedian thalamic stroke results in a hypersomnolent and dearousal state.[7] Contrarily, the predominant involvement of the ventral anterior and the DM nucleus in the neurodegenerative disorder, fatal familial insomnia (FFI) results in chronic sleep deprivation with profound neuropsychological manifestations.[8] The involvement of DM nucleus causing insomnia in FFI compared to hypersomnia in acute strokes has been postulated to be related to the difference in onset (acute vs. gradual), progression (rapid vs. slow), pathological injury (axonal vs. neuronal), and the anatomical localizations of the lesion.[789] In addition, disruption of the Papez circuit involving the anterior and DM nuclei leads to cognitive and the emotional deficits in these patients.[10]
Our patient had a right thalamic stroke leading to a persistent drowsy state with decreased awakening. On 3-month follow-up, he reported significant improvement in his sleep. Bilateral paramedian thalamic infarctions have been well documented to cause hypersomnolence with a dearousal state. However, sleep-wake disturbances following unilateral ischemia are an area of interest.[27]
Bilateral or left thalamic involvement has been shown to predict a poorer recovery when compared to the right thalamic lesions. The poor outcome in the left-sided strokes is associated with lesions of the Papez circuit producing deficits in the frontal lobe-related functions of learning, memory, and language, rather than focal neurological deficits. Right thalamic lesions, on the other hand, have less common cognitive deficits and more complete recovery.[2]
To conclude, thalamic strokes can have protean manifestations ranging from thalamic pain and hemisensory syndrome to excessive sleepiness with attention and cognitive defects. Drowsiness in a stroke patient usually points to large hemispheric strokes with overt neurological deficits. However, in the absence of other focal deficits, it becomes difficult to ascertain the cause of drowsiness or excessive sleepiness to strokes, and most of the evaluation involves testing for metabolic and septic encephalopathies. Hence, it becomes imperative for these patients to undergo neuroimaging to rule out thalamic lesions.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- Evolution of neurological, neuropsychological and sleep-wake disturbances after paramedian thalamic stroke. Stroke. 2008;39:62-8.
- [Google Scholar]
- Cardiovascular physiology: Central and autonomic regulation. In: Kryger MH, Roth T, Dement WC, eds. Principles and Practice of Sleep Medicine. St. Louis, MO: Elsevier Saunders; 2011. p. :215-25.
- [Google Scholar]
- Sympathetic-nerve activity during sleep in normal subjects. N Engl J Med. 1993;328:303-7.
- [Google Scholar]
- Strokes and their relationship with sleep and sleep disorders. Neurologia. 2013;28:103-18.
- [Google Scholar]
- Hypersomnia following paramedian thalamic stroke: A report of 12 patients. Ann Neurol. 1996;39:471-80.
- [Google Scholar]
- Self management of fatal familial insomnia. Part 1: What is FFI? MedGenMed. 2006;8:65.
- [Google Scholar]
- The thalamus and sleep disturbances. In: Guilleminault C, Lugaresi E, Montagna P, Gambetti P, eds. Fatal Familial Insomnia: Inherited Prion Diseases, Sleep and the Thalamus. New York: Raven Press; 1994. p. :177-89.
- [Google Scholar]
- Multiple thalamo-cortical disconnections in anterior thalamic infarction: Implications for thalamic mechanisms of memory and language. Neuropsychologia. 2014;53:264-73.
- [Google Scholar]






