Translate this page into:
Chiari I Malformation Associated with Turner Syndrome
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Turner syndrome (TS) is a rare genetic disease due to the absence of one X chromosome. Patients with TS have more subtle neurological/neuropsychiatric problems, while headache is an uncommon clinical presentation which needs attention. We report a 12-year-old child presenting with typical cough headache. Her magnetic resonance imaging revealed Chiari I malformation associated with TS. To the best of our knowledge, Chiari I malformation associated with TS is not described in literature. We report the first case of TS associated with Chiari I malformation. Interestingly, Chiari I malformation is also associated with Noonan's syndrome, which is a close morphological mimicker of TS, raising the possibility of sharing similar pathogenesis in both conditions.
Keywords
Chiari I malformation
headache
magnetic resonance imaging
Turner syndrome
INTRODUCTION
Turner syndrome (TS) is a rare genetic disease due to the absence of one X chromosome. Neurological manifestations in TS are less frequent. Epilepsy and central nervous system (CNS) tumors account for most of the cases. While neuropsychological abnormalities are more common, they need extensive battery of tests for the identification of specific conditions. Conventional imaging techniques identify gross structural abnormalities, while subtle structural, chemical, and functional abnormalities in the brain are explored by recent advanced techniques in neuroimaging, namely, Voxel-based morphometry (VBM), surface-based morphometry, magnetic resonance spectroscopy (MRS), diffusion tensor imaging (DTI), tractography, and functional magnetic resonance imaging (fMRI). Gross congenital abnormalities are rare and only a few cases are reported in the literature. We describe a first case of Chiari I malformation associated with TS and review about gross and subtle structural, functional, and chemical abnormalities of CNS reported in literature.
CASE REPORT
A 12-year-old female presented with a history of headache of 2 months duration. The headache was in bifrontal region and vertex, more in the early morning, and aggravated with coughing. It started with a bout of respiratory tract infection 2 months back; headache persisted even after the remission of respiratory tract infection. She is only child born to nonconsanguinous parents. There was no history of perinatal insult, cried immediately after delivery. After 2 weeks of birth, she developed respiratory distress. Webbing of the neck was noticed. On evaluation, echocardiogram revealed ventricular septal defect and persistent ductus arteriosus. Her karyotyping disclosed a chromosomal abnormality 46, XO suggestive of TS. She had a history of recurrent respiratory infections and features of cardiac failure during her childhood.
At the time of presentation to our hospital, she had no signs/symptoms of respiratory infection. Recent echo showed pulmonary hypertension. Her oxygen saturation was maintained. On examination, she has short stature, hypertelorism, microcephaly, flat chest, webbing of the neck. She had not attained puberty. She is average in studies.
She underwent MRI of the brain and cervical region, which showed tonsillar herniation, elongation of medulla [Figure 1a], crowding of structures at foramen magnum [Figure 1b], dilated bilateral lateral ventricles, and enlarged third ventricle [Figure 1a and c]. Excessive fat deposition was noted at the nape of neck [Figure 1a]. Focal FLAIR hyperintensity was seen in vermian region, the possible significance of which is not known [Figure 1d]. Cervical vertebral bodies, posterior elements, and spinal cord were normal.
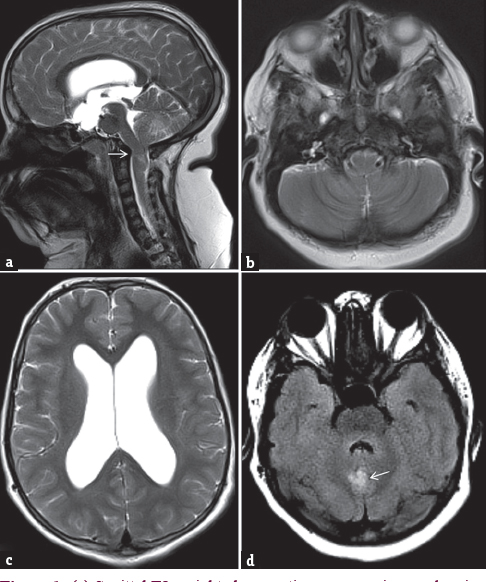
- (a) Sagittal T2-weighted magnetic resonance image showing tonsillar herniation (arrow), elongation of medulla, crowding of structures at foramen magnum, dilated third ventricle. (b) Axial FLAIR magnetic resonance image showing crowding of structures at foramen magnum with effaced cerebrospinal fluid spaces. (c) Axial T2-weighted image showing dilatation of bilateral lateral ventricles. (d) Axial FLAIR image of posterior fossa showing abnormal hyperintense focus in vermian region (arrow)
DISCUSSION
The possibility of CNS abnormalities being part of TS was raised by Brun and Skold in 1968.[1] Neurological symptoms as initial presentation are rare as most of the patients had already diagnosed as TS. Since 1968, multiple structural, functional, and chemical abnormalities of CNS are studied in TS, summarized as follows.
Gross structural abnormalities
Congenital gross structural abnormalities of the CNS in TS detected on conventional imaging modalities are less frequent compared to subtle abnormalities. Five cases of corpus callosum agenesis in association with TS are reported.[23456] A necropsy study from a 45-year-old patient showed an aberrant intracallosal longitudinal fiber bundle that was not detected by conventional MRI.[7] The different MRI abnormalities in patients with epilepsy include bilateral perisylvian hypoplasia,[8] bilateral frontal polymicrogyria,[9] bizarre cortical dysgenesis of the cerebrum – a mixture of relatively normal gyri and structures resembling pachygyria and lissencephaly,[10] diffuse periventricular and subcortical white matter FLAIR hyperintensities.[11] Based on these findings, possible dysgenetic role of X-chromosome was attributed in cortical morphogenesis.
Rare case reports of pseudotumor cerebri,[12] Dandy-Walker malformation, and cerebral venous sinus thrombosis are described.[13] To the best of our knowledge, Chiari I malformation associated with TS is not described in literature. We report the first case of TS associated with Chiari I malformation. Interestingly, Chiari I malformation is also associated with Noonan's syndrome, which is a close morphological mimicker of TS, raising the possibility of sharing similar pathogenesis in both conditions.
Tumors
CNS and non-CNS tumors are seen with increased frequency as compared to general population, suggesting a possible tendency of oncogenesis in TS. Thirteen cases of meningioma associated with TS are described in literature with various neurological presentations including asymptomatic incidental detection.[14] Few cases of meningioma are attributed to estrogen replacement therapy in patients with TS. Few cases of glioblastoma multiforme are seen in associated with TS,[1516] and few cases of craniopharyngioma are reported.[17]
Subtle morphological abnormalities
The subtle neurological deficits in spatial-numerical processing and social cognition seen in patients with TS were attributed to specific alterations of white matter fiber tracts connecting various parts of the brain.[18] Subtle alterations in neurodevelopmental trajectories in some regions of brain were attributed to estrogen deficiency during pre- and post-natal period.[19] Few authors used advanced imaging techniques such as DTI and volumetric imaging techniques to demonstrate extensive aberrant white matter and volume changes in TS.[2021]
Reduced amygdala and hippocampal volumes are also seen which are attributed to social cognition and memory deficits in TS patients.[2223] Reduced volume of corpus callosum, pons, vermis lobules VI–VII and increased fourth ventricle region are reported in TS, suggestive of morphometric abnormalities in posterior fossa structures apart from supratentorial structures.[24] VBM studies showed spatial patterns of altered brain morphometry in TS.[1825] Apart from white matter abnormalities, abnormalities in gray matter (differential decrease/increase in volume of different regions of brain) are also seen using volumetric studies in TS, also contributing to neuropathological basis of cognitive deficiencies.[26272829] Study of surface area of parietal lobe using structural MRI showed an aberrant growth of white matter volume and surface area of the left superior parietal regions in TS.[30] Brain volume analysis using high-resolution MRI also demonstrated decrease in bilateral parietal gray and occipital white matter associated with increased cerebellar gray matter.[31] Studies particularly evaluating superior temporal gyrus abnormalities using volumetric MRI study demonstrated larger volumes of both gray and white matter of right superior temporal gyrus in TS.[3233] Subregional abnormalities within bilateral parietal lobes specifically involving superior parietal and postcentral gyri were seen in TS.[34]
Proton MRS technique was also used to demonstrate abnormalities in chemical composition of the brain parenchyma (lower concentration of N-acetyl aspartate in the lower parietal lobe and higher hippocampal choline).[2735]
TS patients not only have age-independent congenital defect but also have altered brain development, more specifically, age-dependent maturational defect.[36]
Functional abnormalities
Atypical functional activation of the brain was demonstrated during multiple object tracking task,[37] judgment of line orientation task,[38] and Go/NoGo task[39] in patients with TS using fMRI. Visuospatial working memory abnormalities were demonstrated in TS using fMRI studies.[404142]
Cerebrovascular diseases
Several cases of stroke in TS are described, possibly due to vasculopathy/premature atherosclerosis.[43444546] A case of TS presenting with ischemic stroke showed interesting finding of pulvinar sign on T1-weighted imaging of MRI.[47] A rare case of stroke associated with features of fibromuscular dysplasia of the right internal carotid and vertebral arteries was reported.[44]
While dissection of aorta in TS is frequently reported, dissection of carotid-vertebral artery dissection is rare. Few cases of single vessel/multiple cerebral arterial stenosis presenting with stroke are reported.[434548] Few cases of moyamoya syndrome are reported presenting as SAH and intracerebral hemorrhage[49] and stroke-like symptoms.[50] A case of vertebral artery dissection in TS presenting as cervical radiculopathy is reported.[51]
Although few cases of cerebral hemorrhage are reported, hypertension was associated with and possible etiology of such hemorrhages; nevertheless, cerebral vascular abnormalities are also reported in some cases.[52535455]
CONCLUSION
Gross congenital CNS abnormalities are rare, while subtle structural and functional abnormalities are common. Magnetic resonance imaging is useful in identification of abnormalities associated with TS. Conventional MRI needs to be considered in patients with headache to rule out underlying gross structural abnormalities.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- CNS malformations in Turner's syndrome. An integral part of the syndrome? Acta Neuropathol. 1968;10:159-61.
- [Google Scholar]
- Turner's syndrome with agenesis of the corpus callosum, Hashimoto's thyroiditis and horseshoe kidney. Acta Paediatr Jpn. 1987;29:622-6.
- [Google Scholar]
- Ullrich-Turner syndrome with agenesis of the corpus callosum. Am J Med Genet. 1990;37:227-8.
- [Google Scholar]
- Agenesis of the corpus callosum in Turner syndrome with ring X. Dev Med Child Neurol. 1997;39:119-24.
- [Google Scholar]
- Agenesis of the corpus callosum in Turner's syndrome: Report of a case and review of the literature. Acta Neurol Taiwan. 2008;17:194-8.
- [Google Scholar]
- Agenesis of the corpus callosum in a newborn with turner mosaicism. Pediatr Rep. 2014;6:5112.
- [Google Scholar]
- Intracallosal longitudinal fiber bundle: An unexpected finding mimicking demyelination in a patient with Turner syndrome. Acta Neuropathol. 2010;120:545-7.
- [Google Scholar]
- Intractable epilepsy in Turner syndrome associated with bilateral perisylvian hypoplasia: One case report. Clin Neurol Neurosurg. 2005;108:56-9.
- [Google Scholar]
- Bilateral frontal polymicrogyria and epilepsy in a patient with Turner mosaicism: A case report. J Neurol Sci. 2003;213:83-6.
- [Google Scholar]
- Cortical dysgenesis in a patient with Turner mosaicism. Dev Med Child Neurol. 1996;38:455-60.
- [Google Scholar]
- Generalized epilepsy in a patient with mosaic Turner syndrome: A case report. J Med Case Rep. 2014;8:109.
- [Google Scholar]
- Pseudotumour cerebri and the Turner syndrome. J Neurol Neurosurg Psychiatry. 1985;48:164-6.
- [Google Scholar]
- Cerebral venous sinus thrombosis, Dandy-Walker malformation and polymicrogyria in Turner syndrome: An unreported association. Prenat Diagn. 2009;29:899-900.
- [Google Scholar]
- Multiple meningiomas in patients with Turner syndrome. Acta Neurochir (Wien). 2015;157:621-3.
- [Google Scholar]
- Malignant progression of anaplastic supratentorial ganglioglioma into glioblastoma multiforme in a patient with turner syndrome. J Neurol Surg A Cent Eur Neurosurg. 2012;73:253-5.
- [Google Scholar]
- Pediatric glioblastoma multiforme in association with Turner's syndrome: A case report. Pediatr Neurosurg. 2015;50:38-41.
- [Google Scholar]
- An association of craniopharyngioma in Turner syndrome. Pediatr Blood Cancer. 2013;60:E7-9.
- [Google Scholar]
- Brain anatomy in Turner syndrome: Evidence for impaired social and spatial-numerical networks. Cereb Cortex. 2004;14:840-50.
- [Google Scholar]
- Cortical brain morphology in young, estrogen-naive, and adolescent, estrogen-treated girls with Turner syndrome. Cereb Cortex. 2013;23:2159-68.
- [Google Scholar]
- White matter aberrations in prepubertal estrogen-naive girls with monosomic Turner syndrome. Cereb Cortex. 2012;22:2761-8.
- [Google Scholar]
- Aberrant functional network recruitment of posterior parietal cortex in Turner syndrome. Hum Brain Mapp. 2013;34:3117-28.
- [Google Scholar]
- Amygdala and hippocampal volumes in Turner syndrome: A high-resolution MRI study of X-monosomy. Neuropsychologia. 2004;42:1971-8.
- [Google Scholar]
- X-chromosome effects on female brain: A magnetic resonance imaging study of Turnerfs syndrome. Lancet. 1993;342:1197-200.
- [Google Scholar]
- Corpus callosum and posterior fossa development in monozygotic females: A morphometric MRI study of Turner syndrome. Dev Med Child Neurol. 2003;45:320-4.
- [Google Scholar]
- Cognitive impairment and gray/white matter volume abnormalities in pediatric patients with Turner syndrome presenting with various karyotypes. J Pediatr Endocrinol Metab. 2013;26:1111-21.
- [Google Scholar]
- Influence of X chromosome and hormones on human brain development: A magnetic resonance imaging and proton magnetic resonance spectroscopy study of Turner syndrome. Biol Psychiatry. 2006;59:273-83.
- [Google Scholar]
- Cortical thickness correlates of socioemotional difficulties in adults with Turner syndrome. Psychoneuroendocrinology. 2014;44:30-4.
- [Google Scholar]
- Aberrant parietal cortex developmental trajectories in girls with Turner syndrome and related visual-spatial cognitive development: A preliminary study. Am J Med Genet B Neuropsychiatr Genet. 2014;165B:531-40.
- [Google Scholar]
- Brain development in Turner syndrome: A magnetic resonance imaging study. Psychiatry Res. 2002;116:187-96.
- [Google Scholar]
- Effects of X-monosomy and X-linked imprinting on superior temporal gyrus morphology in Turner syndrome. Biol Psychiatry. 2003;54:636-46.
- [Google Scholar]
- Neurodevelopmental effects of X monosomy: A volumetric imaging study. Ann Neurol. 1995;38:731-8.
- [Google Scholar]
- A volumetric study of parietal lobe subregions in Turner syndrome. Dev Med Child Neurol. 2004;46:607-9.
- [Google Scholar]
- Bilateral temporal lobe epilepsy in a patient with Turner syndrome mosaicism. Seizure. 2007;16:261-5.
- [Google Scholar]
- Altered brain development in Turner's syndrome: An event-related potential study. Neurology. 1993;43:801-8.
- [Google Scholar]
- Atypical functional brain activation during a multiple object tracking task in girls with Turner syndrome: Neurocorrelates of reduced spatiotemporal resolution. Am J Intellect Dev Disabil. 2010;115:140-56.
- [Google Scholar]
- Functional neuroanatomy of spatial orientation processing in Turner syndrome. Cereb Cortex. 2004;14:174-80.
- [Google Scholar]
- Abnormal prefrontal cortex function during response inhibition in Turner syndrome: Functional magnetic resonance imaging evidence. Biol Psychiatry. 2003;53:107-11.
- [Google Scholar]
- Functional neuroanatomy of visuo-spatial working memory in Turner syndrome. Hum Brain Mapp. 2001;14:96-107.
- [Google Scholar]
- Reduced functional connectivity during working memory in Turner syndrome. Cereb Cortex. 2011;21:2471-81.
- [Google Scholar]
- Visuospatial executive function in Turner syndrome: Functional MRI and neurocognitive findings. Brain. 2006;129(Pt 5):1125-36.
- [Google Scholar]
- Spontaneous carotid artery dissection in a patient with Turner syndrome. Cerebrovasc Dis. 2007;24:543-4.
- [Google Scholar]
- Cerebral infarction due to stenosis of the bilateral internal carotid artery in Turner's syndrome. No To Shinkei. 1997;49:919-23.
- [Google Scholar]
- Ischemic stroke in a young woman of Turner syndrome with T1-weighted imaging-pulvinar sign. Rinsho Shinkeigaku. 2014;54:440-3.
- [Google Scholar]
- Turner syndrome and occlusion of the internal carotid artery. J Child Neurol. 1993;8:412-5.
- [Google Scholar]
- Moyamoya disease associated with asymptomatic mosaic Turner syndrome: A rare cause of hemorrhagic stroke. J Stroke Cerebrovasc Dis. 2014;23:1242-4.
- [Google Scholar]
- Aortic dissection and moyamoya disease in Turner syndrome. Am J Med Genet A. 2010;152A:2085-9.
- [Google Scholar]
- Vertebral artery dissection in a female patient with Turner's syndrome: Unusual cause of a radicular lesion. Nervenarzt. 2014;85:747-8.
- [Google Scholar]
- Cerebral hemorrhage in turner syndrome: A case report. Clin Pediatr Endocrinol. 2005;14:35-7.
- [Google Scholar]
- A case of aortitis syndrome associated with hypertensive intracranial hemorrhage (author's transl) No To Shinkei. 1982;34:247-52.
- [Google Scholar]
- Putaminal hemorrhage in a case of Turner's syndrome with hyperaldosteronemia. No Shinkei Geka. 2005;33:171-6.
- [Google Scholar]
- Pheochromocytoma and sudden death as a result of cerebral infarction in Turner's syndrome: Report of a case. J Forensic Sci. 1988;33:1497-502.
- [Google Scholar]






